Discrete-PEG® Technology for Precise and Reproducible Conjugation
Introduction
Advances in discrete polyethylene glycol (dPEG) conjugation technology, discrete PEG crosslinkers, and monodisperse PEG crosslinkers have enabled more precise control in antibody‑drug conjugates (ADC), peptide conjugates, nanoparticles, and imaging probes.1 These developments support precision bioconjugation reagents used in therapeutic and diagnostic workflows, where consistency and molecular definition are essential.¹ Polyethylene glycol (PEG) is a key component in bioconjugation and drug formulation, offering biocompatibility, solubility, and epitope shielding to enhance stability, reduce degradation, extend circulation, and lower immunogenicity of therapeutic molecules.2 While conventional PEGs offer beneficial biocompatibility and solubility, their broad molecular weight distribution limits design precision. Despite the utility of conventional PEGs, several challenges persist that limit their effectiveness in bioconjugation.3 Additionally, polydispersity and hydrophobicity can contribute to aggregation and have been linked to immunogenic responses, including anti-PEG antibody formation.4
Vector Laboratories' proprietary dPEG® (discrete PEG) technology addresses these fundamental limitations by providing single-molecular‑weight, highly pure, precision‑engineered PEG linkers with exact chain lengths and orthogonal functional groups.5,6 dPEG®s feature exact chain lengths (such as dPEG4, dPEG12, and dPEG24) with functional groups that include NHS esters, maleimides, azides, carboxylic acids, and amines. These structures enable reproducible and analytically clear bioconjugation for therapeutic and diagnostic applications.⁷ Their controlled synthesis produces consistent homogeneity from 200 Da to 16 kDa, supporting predictable pharmacokinetics and biodistribution. In addition, branched structures can be constructed from three to nine of the linear chains. Discrete PEG reagents have been reported to contain reactive groups, fluorescent probes, metal chelates, drug molecules, affinity ligands, biotin, and many other components. These factors highlight the value of discrete dPEG® linkers as a next‑generation solution for precise and reproducible bioconjugation.
dPEG® Conjugation Technology Overview
dPEG® technology represents a paradigm shift from conventional PEG polymers by introducing molecular precision and design flexibility. The advantages of discrete dPEG® linkers over traditional PEG polymers are summarized in Table 1, including improvements in molecular uniformity, design flexibility, and analytical simplicity.
Figure 1 illustrates the contrast between polydisperse PEG polymers and monodisperse dPEG® crosslinkers, highlighting the single‑molecular‑weight precision achieved through controlled synthesis.
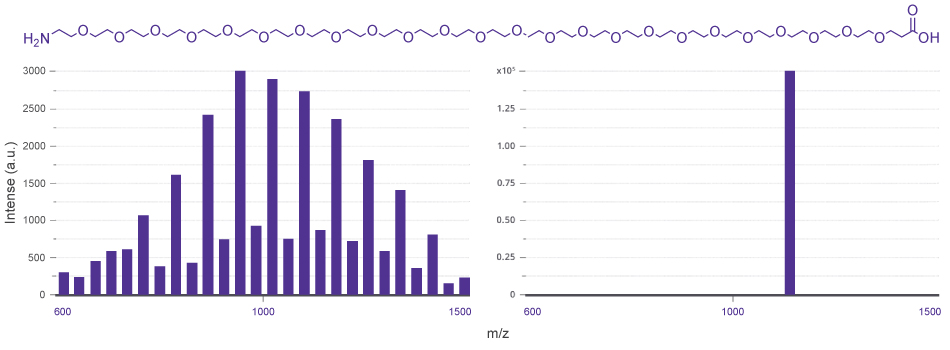
Figure 1.Side by side mass spectra comparing a polydisperse PEG1000 sample (Mw = 1027, Mn = 888, Đ = 1.16; mass ranges between 600–1500 Dalton) with the monodisperse dPEG®24 reagent QBD 10317 (MW =1146.355 Da; Đ =1), showing the shift from dispersed to single molecular weight species.
Vector Labs’ BioDesign™ portfolio includes multiple architectural classes tailored to therapeutic and diagnostic needs:
- Controlled architecture: Precise linear, branched, and orthogonal dPEG® architectures.
- Customizable reactivity: Broad range of reactive groups for flexible conjugation.
- Enhanced hydrophilicity: Superhydrophilic™ spacers for improved solubility and low aggregation.
- Analytical clarity: Defined, monodisperse structures for clean analytics and reproducibility.
The product catalog spans a broad range of applications, including branched dPEG® crosslinkers for high DAR ADCs, Fmoc and t‑Boc protected dPEG® acids for peptide synthesis, azido, alkyne, and DBCO formats for click chemistry, dPEG®‑spaced dyes and chelators for enhanced imaging, and amine or carboxy dPEG® reagents for nanoparticle passivation and reduced nonspecific binding.
dPEG® crosslinkers combine small molecule precision with polymer versatility, enabling rational design in therapeutics, diagnostics, and controlled release systems. With reactive groups such as NHS, maleimide, azide, alkyne, PFP, TFP, haloacetyl, and tetrazine, they support click chemistry, bioorthogonal labeling, enzymatic ligation, and site‑specific conjugation. Their defined architecture improves solubility, reduces aggregation, lowers immunogenicity risk, and delivers reliable bioconjugation performance.
Application Spectrum of dPEG® Technology
The development of dPEG® crosslinkers has broadened the applicability of PEG chemistry across multiple domains of biotechnology, diagnostics, and therapeutic formulation. Their precise structure, modular chemistry, and high hydrophilicity enable reproducible, high-performance conjugations across diverse molecular systems, from peptides and antibodies to nanoparticles and fluorescent dyes. Each listed application leverages the precise architecture and monodispersity of dPEG®, providing clear benefits over traditional heterogeneous PEGs.
Drug Delivery: Liposomes and Nanocarriers
dPEG® crosslinkers are widely used in the formulation of liposome-based drug delivery systems and advanced nanocarriers to address challenges related to solubility, aggregation, and pharmacokinetics.8 Their Superhydrophilic™, monodisperse architecture modulates drug release rates and enhances stability by providing a consistent hydrophilic shell, which reduces opsonization and immune clearance. This controlled surface modification translates into improved bioavailability and longer systemic circulation for encapsulated or conjugated therapeutics, thereby increasing therapeutic efficacy and reducing off-target toxicity.
Serum Half-Life Extension (PEGylation)
Branched and multi-arm dPEG®s serve as molecular spacers and shielders in PEGylation, significantly enhancing the serum half-life of proteins, peptides, and antibody fragments. Unlike traditional polydisperse PEG, discrete PEG architectures prevent batch-to-batch variability and immunogenicity, delivering reproducible pharmacokinetic profiles.9 Clinical evidence shows dPEGylated biotherapeutics resist proteolytic degradation and renal clearance, ensuring sustained therapeutic action for protein drugs, enzymes, and fragments while reducing the risk of anti-PEG antibody formation.
Crosslinkers for IVD/Clinical Conjugates
dPEG® crosslinkers provide versatile architectures for conjugating biomolecules in in vitro diagnostics and clinical assays. Their hydrophilic nature reduces nonspecific binding and aggregation, improving assay sensitivity and reproducibility. These linkers are widely used in ELISA kits, biosensors, and immunoassays where precise molecular spacing and stability are critical for accurate detection.10 Amino-dPEG36-tBu ester was used as a cross-linker in creating well-defined, water-soluble C60-PEG conjugates, which exhibit thermo-responsive properties.11
Fluorescent Labels & Imaging
The application of Superhydrophilic™ dPEG® compounds in fluorescent labels and imaging is transforming antibody and protein labeling. Traditional fluorescent dyes often exhibit high hydrophobicity, leading to antibody instability and increased nonspecific background in assays. In contrast, dPEG® modified fluorescent dyes enhance water solubility and biocompatibility, allowing for more effective labeling without compromising antibody integrity.12 This innovation results in brighter dyes with improved specificity towards targets, enabling greater fluorescence intensity and reducing nonspecific binding in detection applications. Short dPEG® crosslinkers (dPEG4, dPEG8) have been found to improve the performance of antibody-indocyanine green (ICG) optical imaging probes by increasing covalent binding and stability, reducing non-specific background, and enabling higher tumor-to-background imaging ratios in vivo.13
Click Chemistry Reagents
dPEG® click reagents, including azides, alkynes, and DBCO derivatives, enable bioorthogonal conjugation under mild conditions. Their molecular precision ensures predictable reaction kinetics, consistent product profiles, and low immunogenicity.14 Metal-free (strain promoted or copper-free) click ready dPEG®s are particularly valuable for labelling sensitive biomolecules, proteins, or nucleic acids without risking cytotoxic residues.15 Their hydrophilic nature ensures compatibility with aqueous environments, making them ideal for biomedical and nanotechnology applications.
Surface Modifiers
dPEG® crosslinkers and passivators are employed to modify nanoparticles, beads, and sensor surfaces, imparting non-fouling, hydrophilic coatings that greatly suppress nonspecific protein adsorption and cellular uptake.12 Gold and magnetic particles coated with dPEG® exhibit higher targeting specificity and lower “background,” enabling precise biosensing, targeted imaging, and selective cell capture. In addition, dPEG® passivators prolong nanoparticle circulation and stability, critical for diagnostic imaging and drug delivery applications, and help prevent aggregation and opsonization in vivo.
Peptide Building Blocks and Scaffold Assembly
dPEG® serves as a modular scaffold for solid-phase peptide synthesis, enabling the creation of Fmoc, t-boc, thiol, and other crosslinker functionalized building blocks. This flexibility is used to design tailored probes, enzyme substrates, and biotherapeutic conjugates with precise length, hydrophilicity, and reactivity. The resultant constructs feature predictable pharmacokinetic and biological activity, and applications span cancer targeting, enzyme therapy, and imaging agent preparation. Studies have shown building a modular scaffold using discrete-length PEGs for PIE12-trimer synthesis, enabling multimeric D-peptide conjugates with enhanced potency, membrane targeting, and strong resistance to HIV mutations.16
ADC Linker Reagents
ADCs benefit from dPEG® crosslinkers that enhance solubility and reduce aggregation of hydrophobic payloads. These crosslinkers improve pharmacokinetics and therapeutic index by ensuring controlled drug release and minimizing off-target toxicity. dPEG® technology is pivotal in next-generation ADC development for oncology.6 Recent study highlights the role of dPEG® based crosslinkers in ADCs, demonstrating how they provide steric shielding that modulates enzyme-mediated payload release, ultimately enhancing selectivity and stability.17
The dPEG® application spectrum is illustrated in the Table 2 below, linking product types to practical applications.
Find the Right dPEG® Crosslinkers for Your Research Application
dPEG® reagents represent a significant advancement over conventional PEG polymers, offering monodisperse, precisely defined molecular weights and chain lengths. These features provide superior control in bioconjugation, peptide synthesis, drug delivery, and diagnostic applications. Choosing the appropriate dPEG® reactive functional group is critical for optimizing conjugation efficiency, stability, and specificity in your research. The following table correlates common dPEG® reactive end groups with key application areas.
Selection of dPEG® reagents based on this table will enhance experimental reproducibility, specificity, and success in diverse bioconjugation and biomedical applications.
Beyond selecting the right reactive group, choosing the appropriate crosslinker architecture, linear or branched, has a direct impact on ADC developability, stability, solubility, and in vivo performance. Table 4 summarizes how these crosslinker types address common challenges encountered during ADC design.
To further support linker selection, Table 5 compares the key features, benefits, and limitations of linear versus branched dPEG® crosslinkers. This comparison helps researchers determine the most suitable architecture for payload class, desired DAR, stability, and performance requirements.
Outlook for dPEG® Technologies
dPEG® technology is poised to become a cornerstone in next-generation bioconjugation and drug delivery systems. As the demand for precision-engineered linkers grows, dPEG® offers unique advantages that align with emerging trends in biologics, diagnostics, and advanced therapeutics.
Expansion in ADCs and Targeted Therapeutics
The increasing complexity of ADCs and targeted biologics requires crosslinkers that deliver reproducibility, tunable pharmacokinetics, and improved stability. dPEG® architectures, particularly branched and orthogonal designs will enable higher drug-to-antibody ratios without compromising solubility or increasing aggregation, paving the way for safer, more potent ADCs.30
Integration into Peptide and Oligonucleotide Platforms
Peptide-based drugs and oligonucleotide conjugates face challenges of poor solubility and aggregation. Short dPEG® spacers will play a critical role in improving synthesis yields, reducing steric hindrance, and enhancing bioavailability, supporting the growth of peptide therapeutics and RNA-based modalities.31 Several studies have demonstrated altered biodistribution of antibodies or antibody fragments, such as the Fab' fragment (F9) of A5B7 and mCC49 Fab' after PEG modification, resulting in increased tumor accumulation and reduced levels in normal tissues.32
Vaccine and Immunogen Design
Liposomes modified with dPEG® crosslinkers create highly stable and hydrophilic delivery vehicles suitable for encapsulating advanced vaccine modalities (such as mRNA, recombinant proteins, DNA, and cell lysates). Incorporating dPEG® into hapten-carrier conjugates significantly enhances the immune response to the bound antigen or hapten, without triggering immunogenicity toward the linker, an advantage not seen with hydrophobic crosslinkers. This approach is expected to support next-generation vaccines, including mRNA and protein-based platforms.33, 34
Advanced Diagnostics and Imaging
Hydrophilic dPEG® modified dyes, such as AQuora®, already demonstrate superior performance in multiplex imaging and immunostaining. Future developments will extend into PET tracers, real-time in vivo diagnostics, and nanoparticle-based assays, leveraging dPEG® chemistry for enhanced signal clarity and reduced nonspecific binding.5
Addressing PEG Immunogenicity
Reports of anti-PEG antibodies primarily involve high-molecular-weight polymeric PEGs. dPEG®’s discrete, low molecular weight architecture significantly reduces immunogenic risk, positioning it as a safer alternative for long-term therapeutic use.35, 36
Conclusion
dPEG® crosslinkers deliver a level of precision, consistency, and design flexibility that conventional PEGs cannot provide. Their monodisperse architecture supports predictable bioconjugation outcomes, improved solubility, reduced aggregation, and clearer analytical characterization. These advantages are essential for ADC development, peptide and protein conjugation, advanced imaging, nanoparticle engineering, and next‑generation therapeutic design. As bioconjugation workflows increasingly require reproducibility and molecular control, dPEG® technology offers a reliable and high‑performance platform that enables robust, scalable, and future‑ready biologics and diagnostic applications.
Click here to explore our full range of dPEG® products for your applications.
Disclaimer: The Discrete Polyethylene Glycol (dPEG®) technology described here is for R&D use only.