412510 Sigma-AldrichIRE1 Inhibitor I, STF-083010 - Calbiochem
IRE1 Inhibitor I, STF-083010, CAS 307543-71-1, is a cell-permeable compound that directly targets Ireα1 and disrupt Ire1-XBP1 unfolded protein response pathway in RPMI8226 multiple myeloma cells.
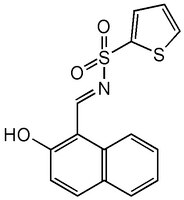
More>> IRE1 Inhibitor I, STF-083010, CAS 307543-71-1, is a cell-permeable compound that directly targets Ireα1 and disrupt Ire1-XBP1 unfolded protein response pathway in RPMI8226 multiple myeloma cells. Less<<Synonyms: N-[(2-Hydroxynaphthalen-1-yl)methylidene]thiophene-2-sulfonamide, N-[(2-Hydroxy-1-naphthyl)methylene]-2-thiophenesulfonamide, ER-to-Nucleus Signaling 1 Inhibitor I, ERN1 Inhibitor I, Inositol-Reguiring Protein 1 Inhibitor I, STF083010
Recommended Products
Overview
| Replacement Information |
|---|
Key Spec Table
| Empirical Formula |
|---|
| C₁₅H₁₁NO₃S₂ |
Products
| Catalogue Number | Packaging | Qty/Pack | |
|---|---|---|---|
| 412510-10MG | Glass bottle | 10 mg |
| References | |
|---|---|
| References | Lerner, A.G., et al. 2012 Cell Metabolism 16, 250. Papandreou, I., et al. 2010. Blood 117, )1311. |
| Product Information | |
|---|---|
| Form | Yellow powder |
| Hill Formula | C₁₅H₁₁NO₃S₂ |
| Chemical formula | C₁₅H₁₁NO₃S₂ |
| Structure formula Image | |
| Quality Level | MQ100 |
| Biological Information | |
|---|---|
| Purity | ≥98% by HPLC |
| Physicochemical Information |
|---|
| Dimensions |
|---|
| Materials Information |
|---|
| Toxicological Information |
|---|
| Safety Information according to GHS |
|---|
| Safety Information |
|---|
| Product Usage Statements |
|---|
| Packaging Information | |
|---|---|
| Packaged under inert gas | Packaged under inert gas |
| Transport Information |
|---|
| Supplemental Information |
|---|
| Specifications |
|---|
| Global Trade Item Number | |
|---|---|
| Catalogue Number | GTIN |
| 412510-10MG | 04055977188165 |
Documentation
IRE1 Inhibitor I, STF-083010 - Calbiochem SDS
| Title |
|---|
IRE1 Inhibitor I, STF-083010 - Calbiochem Certificates of Analysis
| Title | Lot Number |
|---|---|
| 412510 |
References
| Reference overview |
|---|
| Lerner, A.G., et al. 2012 Cell Metabolism 16, 250. Papandreou, I., et al. 2010. Blood 117, )1311. |