Salmonella Contamination and Rapid Detection Methods
Foodborne illness affects public health and development worldwide. Salmonella spp. is a Gram-negative bacterium that is responsible for many foodborne diseases in humans and animals.
Salmonella belongs to Enterobacteriaceae family and there are more than 2600 known serovars (or variations) within the Salmonella species. The serovars of Salmonella are broadly classified into typhoidal and nontyphoidal. The nontyphoidal Salmonella serovars Typhimurium and Enteritidis are two of the most common causes of gastroenteritis, but many others can also lead to illness. As Salmonella is considered a serious threat to human health, the effective identification and rapid detection of Salmonella is crucial to prevent outbreaks of food-borne disease.
Salmonella Contamination in Food
Salmonella bacteria are widely distributed among domestic and wild animals. They are prevalent in animals such as poultry, pigs, and cattle. Salmonella can pass through the entire food chain from animal feed to primary production, and households or food-service establishments and institutions.1
Salmonellosis, the infection caused by Salmonella, can be extremely severe, particularly for young children, elderly and the immuno-compromised. It is generally contracted through the consumption of contaminated food of animal origin (mainly eggs, meat, poultry, and milk), although other foods have also been implicated in its transmission. Symptoms of Salmonellosis include diarrhea, abdominal pain, nausea, and vomiting that can last from 1 to 7 days.
Salmonella can be quite resilient, cannot be killed by freezing, and can also survive in acidic foods. The high resistance to drying, combined with a very high heat resistance once dried, makes Salmonella a potential problem in most foods, particularly in dry and semi-dry products (e.g., milk powder, spices, etc.). This means that if contamination is present, it can be difficult to eradicate. For example, fatty foods can protect the cells from quite severe heat treatments, making pasteurization ineffective.
It is therefore exceptionally important for all food manufacturers to rigorously test for Salmonella to ensure no contaminated products are released to the public.
ISO Method for Salmonella Detection
The International Organization for Standardization (ISO) recently revised the scope of the horizontal method for the detection, enumeration, and serotyping of Salmonella in the revised EN ISO 6579-1:2017 standard which has been expanded to incorporate ISO 6785 I IDF 93 and includes milk and milk products, animal feces and environmental samples from the primary production stage.
For further information, read our article ISO 6579-1 for Salmonella Detection in the Food Chain or watch our 10-minute presentation on compliant Salmonella spp. testing.
Media and ISO Compliance
When following ISO 6579-1:2017, method specific media should be used as the media formulation and preparation should be as described in the method. For confidence during audits or lab inspections requiring ISO adherence, use media that has been released from an ISO/IEC 17025 accredited laboratory, such as our Granucult® dehydrated media, or ready-to-use ReadyTube® media, which also verifies compliance with DIN EN ISO 11133:2014.
Sample Preparation And Enrichment
Sample preparation and pre-enrichment are carried out with buffered peptone water, followed by selective enrichment using Muller-Kauffmann Tetrathionate-Novobiocin (MKTTn) Broth, Rappaport Vasiliadis Soy broth (RVS) broth, or Modified Semi-solid Rappaport Vasiliadis (MSRV) agar.

Discover the new ReadyStream® system that instantly dispenses pre-heated, sterile, and ISO 11133-compliant culture media with the push of a button. The system simplifies media preparation in food testing labs and allows you to prepare up to 100 L of ready-to-use culture media in less than 20 minutes.
Visit our webpage to explore our gravimetric dilutors and sample homogenizers designed to streamline your sample preparation process.
Molecular Methods
Try our rapid and accurate detection method featuring the latest innovations in genetic detection technology and food microbiology. The Assurance® GDS system uses multiple layers of specificity, including a superior sample preparation by using an immunomagnetic separation (IMS) that uses 1 mL test sample volume instead of 100 μL, and that removes efficiently PCR inhibitors, highly specific primers, and a patented probe to ensure highly accurate results. GDS assays have been validated by AOAC and other international standards (AFNOR/MicroVal) according to ISO 16140.
Reference Materials for QC and Validation
To ensure the quality of your test results, it is advisable to use certified reference microorganisms for verification. Our Vitroids™ and LENTICULE® discs contain viable microorganisms in a certified quantity (generally accredited according to ISO/IEC 17025) and are produced under reproducible conditions compliant with ISO Guide 34:2009 using authenticated strains from NCTC®, NCPF®, and CECT®. These discs consist of pure cultures of bacteria in a solid water-soluble matrix, that are stable for at least one year and available in a viable state with a shelf life of 1-3 years. Each batch is provided with a comprehensive certificate of analysis (CoA).

1-2 Test® for Salmonella detection
The 1-2 Test® for Salmonella is an antibody-based assay for the detection of Salmonella in food products, food ingredients and environmental samples. The assay incorporates immunodiffusion technology to deliver accurate and rapid results following a simple 1 or 2-step enrichment protocol. If present, Salmonella moves through the motility media while Salmonella-specific antibodies diffuse into the media from the opposite direction. The antibodies bind to the target Salmonella antigen, forming a visually apparent immunoband that indicates a positive test result. As an AOAC Official Method of Analysis, the 1-2 Test® is one of the simplest rapid methods available for the detection of Salmonella.
The First Widely Accepted Single-Use Salmonella Test
The 1-2 Test® is a patented, single-use test for Salmonella used by analysts worldwide. It combines proprietary science with practical application, resulting in proven performance and ease of use. Each test kit is self-contained and includes all reagents necessary to perform the test. Test results are read visually and are easy to interpret without the use of any additional equipment.
Accurate, Reliable and Validated Results
Extensive multi-laboratory and field validation studies have demonstrated that the 1-2 Test® is highly accurate and specific. The proprietary 1-2 Test® antibody captures a wide range of Salmonella without reacting with most common cross-reactors.
One Test for all Your Samples
The single-use format of the 1-2 Test® provides flexibility to run any number of tests depending on your testing needs. The test requires only two to three minutes of hands-on time to run. Each simple step of the preparation sequence can be performed on an individual unit or simultaneously on multiple units. Results are available as soon as 14 hours after enrichment.
Key Tests/Pathogens: Salmonella
Validations: AOAC OMA 989.13
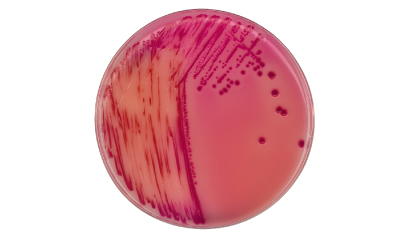
Chromocult® Rambach agar ref. to ISO 6579
Microbiological control in the food industry plays a critical role in preventing Salmonella outbreaks. Tests and media used for the identification of Salmonella take advantage of unique aspects of Salmonella physiology or biochemistry compared to other genera within the family Enterobacteriaceae. For example, bacteria from the genus Salmonella are mostly facultative anaerobes, oxidase-negative, catalase-positive, and gram-negative rods. Most strains are motile and ferment glucose, producing both acid and gas.
Selective Media for Salmonella Detection
EN ISO 6579-1:2017 offers greater flexibility in the plating-out step of Salmonella detection so the procedures for the inoculation of the isolation medium have been made less prescriptive. More detailed directions for the choice of the second isolation medium, which must be used in addition to XLD (Xylose Lysine Deoxycholate) agar, are now given. This medium must be based on different diagnostic characteristics to balance the disadvantages of XLD e.g., the lack of detection of H2S-negative Salmonella. As the second diagnostic agar, we recommend using Rambach® agar, which is based on acid formation with propylene glycol, and in combination with an included pH indicator, the colonies will have a characteristic red color. This enables species of Salmonella to be differentiated unambiguously and allows identification of H2S-negative Salmonella strains, e.g., S. Agona, which may otherwise be missed using XLD agar and a second agar based on the same diagnostic principle, such as XLT4 or Hektoen Enteric Agar.
The media currently used for the differentiation and identification of Salmonella are based on the detection of carbohydrate fermentation, indicated by a pH indicator (see also Table 1 for carbohydrate fermentation abilities), as well as the detection of proteolytic activity, hydrogen sulphide production, and selectivity. Explore the Related Products section for a comprehensive list of the most commonly used selective and differential media.
Table 1, Typical carbohydrate fermentation ability of Salmonella
Chromogenic Media for Faster Salmonella Identification
In addition, our current technology offers chromogenic media, which enhances the reliability and speed of identification by detecting a characteristic enzyme of Salmonella. These reactions are based on the cleavage of a chromogenic substrate, resulting in a visible color change (see Table 2).
Table 2. Chromogenic media for Salmonella
Detection of Hydrogen Sulfide Production of Microorganisms
A large number of bacteria can produce small amounts of H2S from sulfur containing amino acids in carbohydrate media. When H2S is combined with lead acetate, it forms a black precipitate, resulting in a visible black coloured reaction on the paper strip. The lead acetate method is highly sensitive, allowing for the detection of trace levels of hydrogen sulphide.
Test with strips: Inoculate peptone water (Cat. No. 107228) with the suspect organism. Insert a lead acetate paper strip between the plug and inner wall of tube, above the inoculated medium, and incubate at 35 °C for 18-24 hours. A positive reaction appears as a blackening of the lower part of the strip. In the case of negative response, no blackening should appear (see Figure 3).
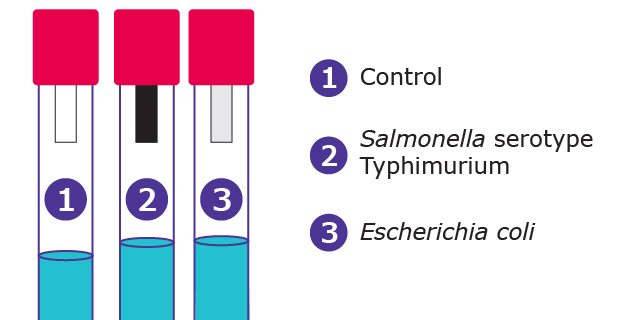
Figure 2: Hydrogen Sulfide Test Strips