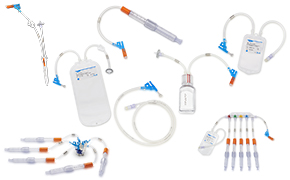
无菌和无菌采样解决方案
一个能够持续产出高质量产物的稳健工艺流程,依赖于可靠的采样方法。在制药和生物制药工艺中实施无菌和无菌采样解决方案,可最大限度降低污染风险,并实现准确、具有代表性的样本采集,从而保障可靠的工艺控制。我们的产品涵盖创新型手动无菌采样和自动化无菌采样解决方案,旨在提升您工艺流程中任何环节的效率与可靠性。
手动无菌采样
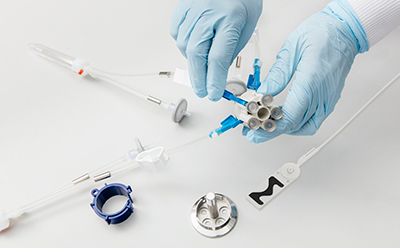
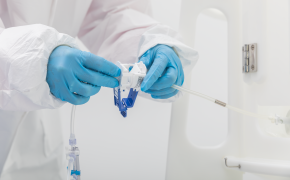
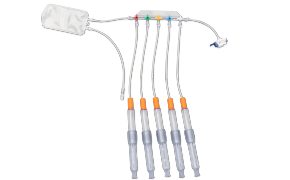
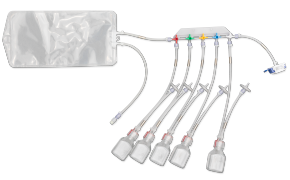
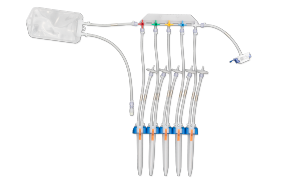
封闭式无菌采样可在生产过程中为您提供所需的安全保障。NovaSeptum® GO无菌采样系统和NovaSeptum® SURe无菌采样组件作为封闭式采样解决方案,能显著降低交叉污染风险,确保在整个生产过程中获取具有代表性的样本。 解决方案包括适用于一次性生产工艺的NovaSeptum® SURe无菌采样组件,以及可连接任何不锈钢单元操作的NovaSeptum® GO无菌采样系统。
- 封闭式设计:保障工艺与样本完整性
- 操作便捷:提供多种连接选项及容器规格的即插即用方案
探索适用于不锈钢或多用途工艺的NovaSeptum® GO系统
自动化无菌采样
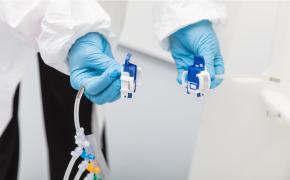
自动化采样系统可实现高频次测量,在最大化效率的同时最小化操作人员干预,适用于上游与下游工艺。我们的MAST®自动采样解决方案支持在线近实时分析,助力全面掌握工艺状况并优化过程控制。
- 高效:提高采样频率,缩短周转时间,最大限度减少操作员接触点
- 可靠:确保快速样品转移与可追溯性,同时维持样品源无菌状态
Products
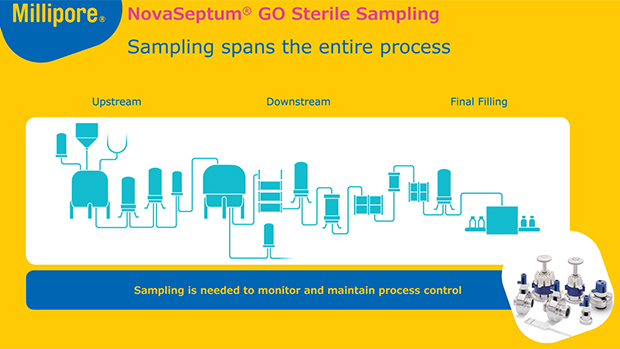
无菌生产流程每个环节的可靠采样
从生物反应器到最终灌装,NovaSeptum®无菌采样系统为生物制药企业提供稳定且具有代表性的样本,用于分析pH值、电导率、细胞活力、代谢物,监测生物负荷及内毒素水平。
通过采样监测pH值、渗透压和/或电导率,并据此调整工艺参数。
启用无菌流体添加功能,以调节或为您的工艺提供物料。
所有采样容器均能实现微生物的卓越回收率,配备在线连接支架,便于添加额外的生物负荷监测步骤。
轻松监测生物负荷、内毒素及质量,确保在下一加工步骤前有效保护昂贵的树脂免受污染物侵害。
用于验证无菌过滤有效性的样品,经验证的采样容器确保不影响内毒素回收率,最大限度减少宝贵产品的样品用量。
无缝集成至最终灌装流程,高安全等级,具有代表性的样本。
为您的工艺选择最佳一次性采样解决方案时,首先需考虑首选连接类型:热熔焊接或无菌连接。预配置组件可通过C-Flex®软管或AsepticQuik®连接器轻松集成至您的工艺流程。
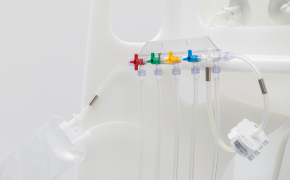
根据样本体积、检测类型、储存条件以及采样计划中的其他关键参数,选择合适的采样容器。我们的NovaSeptum® SURe无菌采样组件提供标准单容器和三/五容器多路分流单元,可选配袋式、瓶式、注射器式或锥形管式容器。
选择适合您工艺的夹具:三夹头(TC)、直通式或In-Gold®。TC夹具还提供预装多种容器类型的选项。
提供多种规格的瓶、袋、注射器和锥形管供您选择。这些产品涵盖多种尺寸和配置——包括单采样、多路采样器或预装式。
确保整个过程保持无菌状态。使用Novaseptum®手动压接工具断开样本连接。
为最大限度提升使用便捷性,我们提供多种配件,包括行李架、端口塞和防盗标签。

MAST®自动采样解决方案是一款先进的自动化无菌采样工具,旨在提升生物制药工艺的效率与控制水平。 该多功能解决方案支持从多个源头直接采集样品并输送至各类分析目的地,从而加速周转时间、减少操作接触点并最大限度降低污染风险。它可与其他过程分析技术(PAT)工具无缝集成以实现全面监测,并支持灵活配置以适应不同工艺流程和规模。作为符合GMP标准的解决方案,它在确保严格合规的同时,提供关键工艺参数的在线近实时分析及高效资源利用。
相关视频
相关产品资源
- White Paper: Aseptic Process Sampling Risk Mitigation – A Regulatory Perspective
There are significant consequences associated with microbial contamination during biopharmaceutical manufacturing. Contamination increases risks for the operator, the company, and potentially the patient, all of which can result in significant negative impact.
- Application Note: Microbial Integrity of NovaSeptum® Sampling Systems upon Performance of Multiple Sampling Actuations
The NovaSeptum® and NovaSeptum® GO sampling systems are a family of products designed for singleuse sterile sampling throughout the biomanufacturing process.
- User Guide: NovaSeptum® and NovaSeptum® GO Sampling Units
This user guide provides installation and use instructions
- Datasheet: NovaSeptum® GO Sterile Sampling System
To demonstrate the safety and integrity of your product, you need a standard-setting sampling solution that provides the flexibility to sample throughout your entire process.
- Datasheet: NovaSeptum® SURe Sterile Sampling Assemblies
NovaSeptum® SURe single-use sterile sampling assemblies are designed to protect your single-use process from potential cross-contamination ensuring representative samples.
- Brochure: NovaSeptum® GO Sterile Sampling System
Continuing to set the sampling standard, our NovaSeptum® GO sterile sampling system is equipped with additional features for even safer sampling throughout your entire process.
- Technical article: Enhance Bioprocess Sampling with MAST® Autosampling Solution
Explore the MAST® Autosampling Solution for efficient, automated sampling in bioprocesses, supporting higher measurement frequency and reduced process risk.
- Datasheet: MAST® Autosampling Solution
The MAST® Autosampling Solution is a versatile & modality-agnostic aseptic autosampling solution for reliable on-line and near real-time sample analysis.
- White Paper: Automated Aseptic Sampling for Accelerated Access to Process and Quality Data in Upstream Bioprocessing
This white paper focuses on PAT and the use of automated sampling technology to accelerate analytical and quality control methods and provide an approach for access to in-line data to monitor processes in real time.
- Application Note: Enabling Accelerated Raman Model Calibration for Seamless and Reproducible Real-Time Monitoring by Combining Raman and Automated Sampling Technologies
Explore how automated sampling can accelerate the integration of a Raman analyzer for a bioreactor application, speeding up the Raman model building phase, and facilitating in-line, real-time monitoring of critical process parameters (CPPs) and critical quality attributes (CQAs) during the manufacturing process.
- White Paper: Accelerate Process Development with Automated Aseptic Sampling
This whitepaper describes evaluation of the MAST® Autosampling Solution as part of an automated PAT system implemented by Takeda Pharmaceuticals.
如要继续阅读,请登录或创建帐户。
暂无帐户?