一次性设备与过滤器验证服务
选择合适的验证服务
我们的验证项目经理精通全球及本地法规与行业最佳实践,助您选择合适的验证服务。我们可协助制定并实施恰当的验证策略——确定测试内容、方法及时间节点,涵盖上游、下游或最终灌装操作。
探索更轻松的选择验证服务之道
向上游的配料罐/混合袋
- 化学兼容性
- 可萃取物/患者安全
最终储存一次性组件
- 化学兼容性
- 可萃取物/患者安全/可浸出物
- 非渗透性测试
最终灭菌过滤器和/或一次性组件
- 细菌截留验证
- 气泡点/扩散测定
- 化学兼容性
- 可萃取物/患者安全性/可浸出物
- 结合研究
- 颗粒脱落研究
- Vmax™确认(即滤膜堵塞评估)
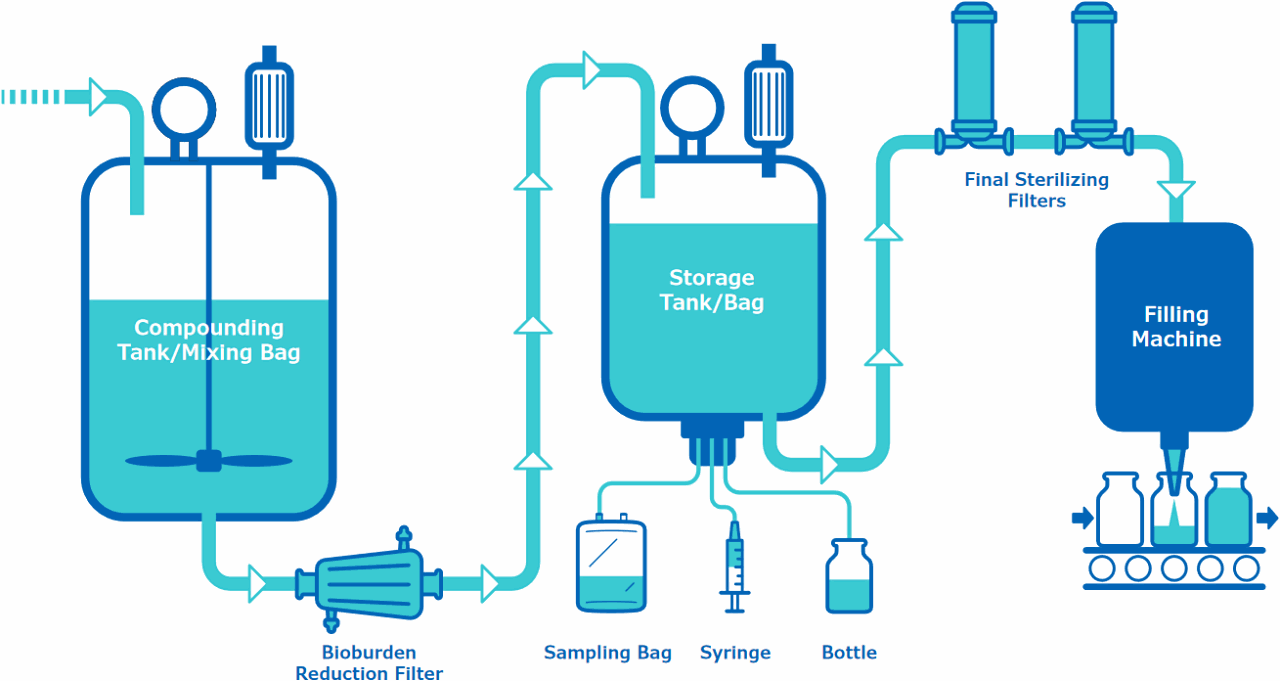
生物负荷降低过滤器
- 化学兼容性
- 可萃取物/患者安全
- 气泡点/扩散测定
采样系统
- 化学兼容性
- 功能性测试
- 防渗漏测试
监管指南
基于药品申请和风险评估的建议
信赖我们的全球服务网络
作为我们为生物制药制造商提供的行业领先验证服务的一部分,我们在全球实验室提供这些专业能力。
- 细菌截留测试
- 化学兼容性测试
- 滤膜完整性测试
- 可萃取物与可浸出物
- 验证服务咨询
- 验证服务等级
相关产品资源
- Validation Services Extractables and Leachables Testing Brochure
This brochure provides details on our validation services for extractables and leachables.
- Validation Services Global Brochure
Regulatory guidance documents provide a framework to manufacturers for the production of drugs that are safe for administration to patients.
- App Note: When Do You Need to Consider Revalidating the Performance of Your Sterilizing-grade Filter?
Any product or process change that could potentially affect the cGMP compliance of a validated process or system should be evaluated and a risk assessment should be performed to evaluate the impact.
- Aseptic Validation Master Plan
Our experienced Validation Services team has summarized best practices for validating performance of critical filtration systems used in aseptic processing. If you need help, they can provide support on the various tests to meet global regulatory requirements.
- Process Development and Drug Manufacturing: Support Services
We provide comprehensive services for drug development and manufacturing, including technical and regulatory expertise and process development support.
