
Bioburden Testing
Reliable Bioburden Testing
|
Request Information |
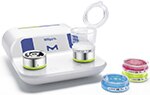
Milliflex Oasis® Filtration System
Our state-of-the-art bioburden testing system brings unmatched comfort to your lab by raising productivity, result reliability and regulatory compliance to a new level.

Milliflex® Quantum Rapid Detection
The Milliflex® Quantum system is compliant for the pharma industry and uses industry-standard membrane filtration techniques and a rapid enzymatic fluorescent staining method for convenient quantitative detection of viable and culturable microorganisms in filterable samples.

EZ Fluo™ Rapid Detection
The EZ-Fluo™ Rapid Detection System is an easy-to-use, non-destructive, fluorescent staining-based system for rapid detection and quantification of microbial contamination in lower-risk samples like food & beverages.

Ready-to-Use Culture Media for Bioburden Testing
For bioburden testing (including microbial limit testing) of non-sterile raw materials, water and finished products, Merck provides all the culture media required to detect viable aerobic microorganisms. The ready–to-use solid and liquid media are prepared according to the corresponding, harmonized standards of the European, US and Japanese Pharmacopeias.

Microbiology Services
Find all you need to benefit from our comprehensive services for bioburden testing including service plans, and regulatory and technological expertise.








