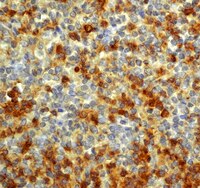
Histone deacetylase expression in white matter oligodendrocytes after stroke.
Kassis, H; Chopp, M; Liu, XS; Shehadah, A; Roberts, C; Zhang, ZG
Neurochemistry international
77
17-23
2014
Show Abstract
Histone deacetylases (HDACs) constitute a super-family of enzymes grouped into four major classes (Class I-IV) that deacetylate histone tails leading to chromatin condensation and gene repression. Whether stroke-induced oligodendrogenesis is related to the expression of individual HDACs in the oligodendrocyte lineage has not been investigated. We found that 2 days after stroke, oligodendrocyte progenitor cells (OPCs) and mature oligodendrocytes (OLGs) were substantially reduced in the peri-infarct corpus callosum, whereas at 7 days after stroke, a robust increase in OPCs and OLGs was observed. Ischemic brains isolated from rats sacrificed 7 days after stroke were used to test levels of individual members of Class I (1 and 2) and Class II (4 and 5) HDACs in white matter oligodendrocytes during stroke-induced oligodendrogenesis. Double immunohistochemistry analysis revealed that stroke substantially increased the number of NG2+OPCs with nuclear HDAC1 and HDAC2 immunoreactivity and cytoplasmic HDAC4 which were associated with augmentation of proliferating OPCs, as determined by BrdU and Ki67 double reactive cells after stroke. A decrease in HDAC1 and an increase in HDAC2 immunoreactivity were detected in mature adenomatous polyposis coli (APC) positive OLGs, which paralleled an increase in newly generated BrdU positive OLGs in the peri-infarct corpus callosum. Concurrently, stroke substantially decreased the acetylation levels of histones H3 and H4 in both OPCs and OLGs. Taken together, these findings demonstrate that stroke induces distinct profiles of Class I and Class II HDACs in white matter OPCs and OLGs, suggesting that the individual members of Class I and II HDACs play divergent roles in the regulation of OPC proliferation and differentiation during brain repair after stroke. | | | 24657831
 |
Collagen signaling enhances tumor progression after anti-VEGF therapy in a murine model of pancreatic ductal adenocarcinoma.
Aguilera, KY; Rivera, LB; Hur, H; Carbon, JG; Toombs, JE; Goldstein, CD; Dellinger, MT; Castrillon, DH; Brekken, RA
Cancer research
74
1032-44
2014
Show Abstract
There is growing evidence that antiangiogenic therapy stimulates cancer cell invasion and metastasis. However, the underlying molecular mechanisms responsible for these changes have not been fully defined. Here, we report that anti-VEGF therapy promotes local invasion and metastasis by inducing collagen signaling in cancer cells. We show that chronic VEGF inhibition in a genetically engineered mouse model of pancreatic ductal adenocarcinoma (PDA) induces hypoxia, a less differentiated mesenchymal-like tumor cell phenotype, TGF-β expression, and collagen deposition and signaling. In addition, we show that collagen signaling is critical for protumorigenic activity of TGF-β in vitro. To further model the impact of collagen signaling in tumors, we evaluated PDA in mice lacking Sparc, a protein that reduces collagen binding to cell surface receptors. Importantly, we show that loss of Sparc increases collagen signaling and tumor progression. Together, these findings suggest that collagen actively promotes PDA spread and that enhanced disease progression associated with anti-VEGF therapy can arise from elevated extracellular matrix-mediated signaling. | | | 24346431
 |
The expression of spinal methyl-CpG-binding protein 2, DNA methyltransferases and histone deacetylases is modulated in persistent pain states.
Tochiki, KK; Cunningham, J; Hunt, SP; Géranton, SM
Molecular pain
8
14
2012
Show Abstract
DNA CpG methylation is carried out by DNA methyltransferases and induces chromatin remodeling and gene silencing through a transcription repressor complex comprising the methyl-CpG-binding protein 2 (MeCP2) and a subset of histone deacetylases. Recently, we have found that MeCP2 activity had a crucial role in the pattern of gene expression seen in the superficial dorsal horn rapidly after injection of Complete Freund's Adjuvant (CFA) in the rat ankle joint. The aim of the present study was to analyse the changes in expression of MeCP2, DNA methyltransferases and a subset of histone deacetylases in the superficial dorsal horn during the maintenance phase of persistent pain states. In this process, the cell specific expression of MeCP2 was also investigated.Using immunohistochemistry, we found that neurones, oligodendrocytes and astrocytes expressed MeCP2. Microglia, oligodendrocyte precursor cells and Schwann cells never showed any positive stain for MeCP2. Quantitative analyses showed that MeCP2 expression was increased in the superficial dorsal horn 7 days following CFA injection in the ankle joint but decreased 7 days following spared nerve injury. Overall, the expression of DNA methyltransferases and a subset of histone deacetylases followed the same pattern of expression. However, there were no significant changes in the expression of the MeCP2 targets that we had previously shown are regulated in the early time points following CFA injection in the ankle joint. Finally, the expression of MeCP2 was also down regulated in damaged dorsal root ganglion neurones following spared nerve injury.Our results strongly suggest that changes in chromatin compaction, regulated by the binding of MeCP2 complexes to methylated DNA, are involved in the modulation of gene expression in the superficial dorsal horn and dorsal root ganglia during the maintenance of persistent pain states. | | | 22369085
 |
Sensory and cortical activation of distinct glial cell subtypes in the somatosensory thalamus of young rats.
H Rheinallt Parri,Timothy M Gould,Vincenzo Crunelli
The European journal of neuroscience
32
2010
Show Abstract
The rodent ventrobasal (VB) thalamus receives sensory inputs from the whiskers and projects to the cortex, from which it receives reciprocal excitatory afferents. Much is known about the properties and functional roles of these glutamatergic inputs to thalamocortical neurons in the VB, but no data are available on how these afferents can affect thalamic glial cells. In this study, we used combined electrophysiological recordings and intracellular calcium ([Ca(2+)](i)) imaging to investigate glial cell responses to synaptic afferent stimulation. VB thalamus glial cells can be divided into two groups based on their [Ca(2+)](i) and electrophysiological responses to sensory and corticothalamic stimulation. One group consists of astrocytes, which stain positively for S100B and preferentially load with SR101, have linear current-voltage relations and low input resistance, show no voltage-dependent [Ca(2+)](i) responses, but express mGluR5-dependent [Ca(2+)](i) transients following stimulation of the sensory and/or corticothalamic excitatory afferent pathways. Cells of the other glial group, by contrast, stain positively for NG2, and are characterized by high input resistance, the presence of voltage-dependent [Ca(2+)](i) elevations and voltage-gated inward currents. There were no synaptically induced [Ca(2+)](i) elevations in these cells under control conditions. These results show that thalamic glial cell responses to synaptic input exhibit different properties to those of thalamocortical neurons. As VB astrocytes can respond to synaptic stimulation and signal to neighbouring neurons, this glial cell organization may have functional implications for the processing of somatosensory information and modulation of behavioural state-dependent thalamocortical network activities. Full Text Article | | | 20608967
 |
A powerful transgenic tool for fate mapping and functional analysis of newly generated neurons.
Zhang, J; Giesert, F; Kloos, K; Vogt Weisenhorn, DM; Aigner, L; Wurst, W; Couillard-Despres, S
BMC neuroscience
11
158
2010
Show Abstract
Lack of appropriate tools and techniques to study fate and functional integration of newly generated neurons has so far hindered understanding of neurogenesis' relevance under physiological and pathological conditions. Current analyses are either dependent on mitotic labeling, for example BrdU-incorporation or retroviral infection, or on the detection of transient immature neuronal markers. Here, we report a transgenic mouse model (DCX-CreERT2) for time-resolved fate analysis of newly generated neurons. This model is based on the expression of a tamoxifen-inducible Cre recombinase under the control of a doublecortin (DCX) promoter, which is specific for immature neuronal cells in the CNS.In the DCX-CreERT2 transgenic mice, expression of CreERT2 was restricted to DCX+ cells. In the CNS of transgenic embryos and adult DCX-CreERT2 mice, tamoxifen administration caused the transient translocation of CreERT2 to the nucleus, allowing for the recombination of loxP-flanked sequences. In our system, tamoxifen administration at E14.5 resulted in reporter gene activation throughout the developing CNS of transgenic embryos. In the adult CNS, neurogenic regions were the primary sites of tamoxifen-induced reporter gene activation. In addition, reporter expression could also be detected outside of neurogenic regions in cells physiologically expressing DCX (e.g. piriform cortex, corpus callosum, hypothalamus). Four weeks after recombination, the vast majority of reporter-expressing cells were found to co-express NeuN, revealing the neuronal fate of DCX+ cells upon maturation.This first validation demonstrates that our new DCX-CreERT2 transgenic mouse model constitutes a powerful tool to investigate neurogenesis, migration and their long-term fate of neuronal precursors. Moreover, it allows for a targeted activation or deletion of specific genes in neuronal precursors and will thereby contribute to unravel the molecular mechanisms controlling neurogenesis. | Immunohistochemistry | | 21194452
 |
In vivo intracellular recording suggests that gray matter astrocytes in mature cerebral cortex and hippocampus are electrophysiologically homogeneous.
Mishima, T; Hirase, H
The Journal of neuroscience : the official journal of the Society for Neuroscience
30
3093-100
2010
Show Abstract
Previous anatomical and in vitro electrophysiology studies suggest that astrocytes are heterogeneous in physiology, morphology, and biochemical content. However, the extent to which this diversity applies to in vivo conditions is largely unknown. To characterize and classify the physiological and morphological properties of cerebral cortical and hippocampal astrocytes in the intact brain, we performed in vivo intracellular recordings from single astrocytes using anesthetized mature rats. Astrocytes were classified based on their glial fibrillary acidic protein (GFAP) immunoreactivity and cell body locations. We analyzed morphometric measures such as the occupied volume and polarity, as well as physiological characteristics such as the mean membrane potential. These measurements did not show obvious segregation into subpopulations, suggesting that gray matter astrocytes in the cortex and hippocampus are composed of a homogeneous population in mature animals. The membrane potential of astrocytes in both cortex and hippocampus fluctuated within a few millivolts in the presence of spontaneous network activity. These membrane potential fluctuations of an astrocyte showed a significant variability that depended on the local field potential state and cell body location. We attribute the variability of the membrane potential fluctuations to local potassium concentration changes due to neuronal activity. | Immunohistochemistry | | 20181606
 |
JAM-A is a novel surface marker for NG2-Glia in the adult mouse brain.
Stelzer, Sandra, et al.
BMC Neurosci, 11: 27 (2010)
2010
Show Abstract
BACKGROUND: Junctional adhesion molecule-A (JAM-A) is an adhesive protein expressed in various cell types. JAM-A localizes to the tight junctions between contacting endothelial and epithelial cells, where it contributes to cell-cell adhesion and to the control of paracellular permeability. RESULTS: So far, the expression pattern of JAM-A has not been described in detail for the different cell types of the adult brain. Here we show that a subset of proliferating cells in the adult mouse brain express JAM-A. We further clarify that these cells belong to the lineage of NG2-glia cells. Although these mitotic NG2-glia cells express JAM-A, the protein never shows a polarized subcellular distribution. Also non-mitotic NG2-glia cells express JAM-A in a non-polarized pattern on their surface. CONCLUSIONS: Our data show that JAM-A is a novel surface marker for NG2-glia cells of the adult brain. | | | 20184779
 |
Genesis of neuronal and glial progenitors in the cerebellar cortex of peripuberal and adult rabbits.
Ponti, G; Peretto, P; Bonfanti, L
PloS one
3
e2366
2008
Show Abstract
Adult neurogenesis in mammals is restricted to some brain regions, in contrast with other vertebrates in which the genesis of new neurons is more widespread in different areas of the nervous system. In the mammalian cerebellum, neurogenesis is thought to be limited to the early postnatal period, coinciding with end of the granule cell genesis and disappearance of the external granule cell layer (EGL). We recently showed that in the rabbit cerebellum the EGL is replaced by a proliferative layer called 'subpial layer' (SPL) which persists beyond puberty on the cerebellar surface. Here we investigated what happens in the cerebellar cortex of peripuberal rabbits by using endogenous and exogenously-administered cell proliferation antigens in association with a cohort of typical markers for neurogenesis. We show that cortical cell progenitors extensively continue to be generated herein. Surprisingly, this neurogenic process continues to a lesser extent in the adult, even in the absence of a proliferative SPL. We describe two populations of newly generated cells, involving neuronal cells and multipolar, glia-like cells. The genesis of neuronal precursors is restricted to the molecular layer, giving rise to cells immunoreactive for GABA, and for the transcription factor Pax2, a marker for GABAergic cerebellar interneuronal precursors of neuroepithelial origin that ascend through the white matter during early postnatal development. The multipolar cells are Map5+, contain Olig2 and Sox2 transcription factors, and are detectable in all cerebellar layers. Some dividing Sox2+ cells are Bergmann glia cells. All the cortical newly generated cells are independent from the SPL and from granule cell genesis, the latter ending before puberty. This study reveals that adult cerebellar neurogenesis can exist in some mammals. Since rabbits have a longer lifespan than rodents, the protracted neurogenesis within its cerebellar parenchyma could be a suitable model for studying adult nervous tissue permissiveness in mammals. | | Rabbit | 18523645
 |
Multiple regions of the NG2 proteoglycan inhibit neurite growth and induce growth cone collapse
Ughrin, Yvonne M, et al
J Neurosci, 23:175-86 (2003)
2003
| Immunohistochemistry (Tissue) | Rat | 12514214
 |
The oligodendrocyte precursor cell in health and disease
Levine, J M, et al
Trends Neurosci, 24:39-47 (2001)
2001
| | | 11163886
 |