
Bioburden Testing for Pharmaceutical & Cosmetics
Bioburden Testing Solutions |
Request Information |
For bioburden testing in the pharmaceutical and cosmetics industries, we provide a complete portfolio of products and services designed to ensure microbiologically safe drugs and cosmetics. Our products range from traditional to rapid and are based on proven technologies such as membrane filtration and fluorescence-based, non-destructive detection.
Milliflex Oasis® Filtration System
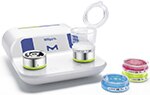 Our state-of-the-art bioburden testing system brings unmatched comfort to your lab by raising productivity, result reliability and regulatory compliance to a new level.
Our state-of-the-art bioburden testing system brings unmatched comfort to your lab by raising productivity, result reliability and regulatory compliance to a new level.
The Complete Range of Media Casettes
 For bioburden testing, we maintain a complete range of agar-based media in convenient, pre-filled, ready-to-use cassettes. Our high purity ingredients and ISO-certified manufacturing processes give our media a longer shelf life than formulations prepared on-site. We extensively test each lot of media for pH, sterility assurance, recovery of ATCC strains typically found in bioburden applications, and product formulation. A Certificate of Quality containing all relevant test data is included in every box. Our media conform to the requirements set forth in the European, US and Japanese pharmacopeias.
For bioburden testing, we maintain a complete range of agar-based media in convenient, pre-filled, ready-to-use cassettes. Our high purity ingredients and ISO-certified manufacturing processes give our media a longer shelf life than formulations prepared on-site. We extensively test each lot of media for pH, sterility assurance, recovery of ATCC strains typically found in bioburden applications, and product formulation. A Certificate of Quality containing all relevant test data is included in every box. Our media conform to the requirements set forth in the European, US and Japanese pharmacopeias.
The Rapid Testing Alternative Milliflex® Quantum
 Be the first to learn how easy it really is to use the Milliflex® Quantum rapid system in your lab to detect microbial contamination faster, improving your process control. Ideally suited for testing raw materials, water and drug products.
Be the first to learn how easy it really is to use the Milliflex® Quantum rapid system in your lab to detect microbial contamination faster, improving your process control. Ideally suited for testing raw materials, water and drug products.
- It’s non-destructive: you can easily recover and collect any microorganisms detected for identification using existing ID technology.
- It’s easy to use: with a simple protocol, the hardware and a workflow, it only takes a few hours to familiarize yourself with the system and begin using it. Validation is streamlined due to limited hardware needing to be used.
- Results compare with those of traditional methods: being based on compendial methods using our proven Milliflex Oasis® funnels, the Milliflex® Quantum system requires few additional steps. Sample preparation and incubation conditions are identical to traditional microbiological methods, as is the reporting of actual membrane filtration, which facilitates the validation process.
- Faster than traditional methods: the microbiological results are available in a fraction of the time that traditional methods take, allowing you to respond to contamination earlier in the process and, ultimately, save time and money.






