

Introducing our Plug & Play Upstream Development Service for Small-sized Biotechs
Adaptive solutions that help you get from DNA to clinic, the right way for you
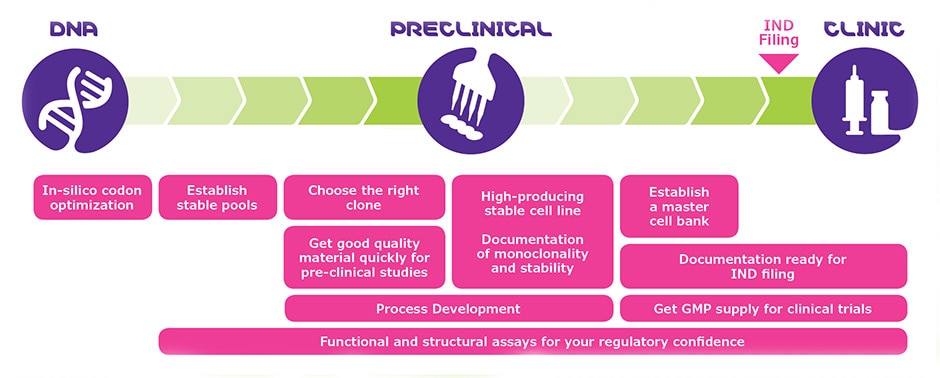
Read our article and find out what are the critical steps for small-sized biotechs to get through the preclinical phase quickly and cost-effectively.
Merck Introduces New Integrated Plug & Play Upstream Development Service
Read our press release
Our innovative, custom approach helps small biotechs accelerate their upstream development: Here’s how we do it
Based on our automated mini-pool approach
Learn how this approach allows to efficiently create, screen and isolate high producing clones with reduced cost and time. Other approaches will be described in this case study, such as cell type selection, expression cassette design and super-transfection.
Based on a multiparallel workflow process
Learn from BDS Team Leader, Cyrielle Corbin, Ph.D. about our mini-pool approach as fast-track process and how we can begin process development in parallel with cell line development, reducing timelines by 10-13 weeks.

Our innovative, custom approach helps small biotechs find nimble solutions for their upstream development: Here’s how we do it
Based on a super-transfection approach
A customer sought to increase the titer of a molecule that had proven difficult to express, with a titer insufficient for clinical studies. Learn how our strategy allowed to increase the titer nearly 4-fold after generation of stable pools.
Based on a 200 Liter scale
Learn how our customers reduce cost and time by leveraging our CHOZN® cell line and semi-automated approach to mini-pools and reach a 200L scale.
A service designed around your needs - choose the path that’s right for you
Choose our proprietary CHOZN® platform, which allows for faster, simpler selection and scale-up of high-producing clones.
We help you find the right balance between speed and efficiency, so your clone demonstrates high quality and productivity early on to avoid delays in your clinical trials.
Leverage our innovative approach to mini-pools and confidently run process development and clone selection in parallel and save up to 10 weeks.
Run analytical development in parallel to your cell line development to get you progressing faster. Process transfer between our teams is seamless.
One Tailored Suite of Upstream Development Services
A plug & play service offering, exaclty tailored to small-sized biotechs, needing to balance cost, risk and speed to clinic without sacrificing quality
- Full cell line development services from your molecule of interest to the Research Cell Bank of the clones
- Validated cell line development process based on mini-pool approach and QA validated limit dilution cloning
- Variants and signal peptides screening possible during early phases of cell line development
- Monoclonality reports and stability validation in compliance with regulatory expectations
- Add-on services for Analytical method development and product characterization
- Fast-Track process using the mini-pool as early material for process development saving 10-13 weeks on overall project timeline
Ask our Experts
Learn from BDS Scientist Clémence Justine about factors that must be considered when optimizing recombinant antibody expression.
Learn from Research BDS Scientist Lucie Grinders about our mini-pool approach and how we can begin process development in parallel with cell line development.
Learn from BDS Team Leader, Cyrielle Corbin, Ph.D. how we run steps in parallel for customers who have a very challenging timeline.
Learn from Senior Scientist Helen Hsu, Ph.D. how, with our custom binding assays, and supported with our comprehensive off-the-shelf Fc gamma receptor binding assays, we optimize your mAb production.
Our Experience

BioReliance® End-to-End Solutions
We are an adaptable CDMO partner for start-ups and small biotechs needing to develop and commercialize biologics. We do this by balancing speed, risk and cost through custom solutions, by leveraging our bioprocessing technologies and process development expertise, and by allowing our clients to transfer their process and knowledge to their end point at any step of their drug development. To learn more, please visit BioReliance® End-to-End Solutions - Your CDMO Partner.










